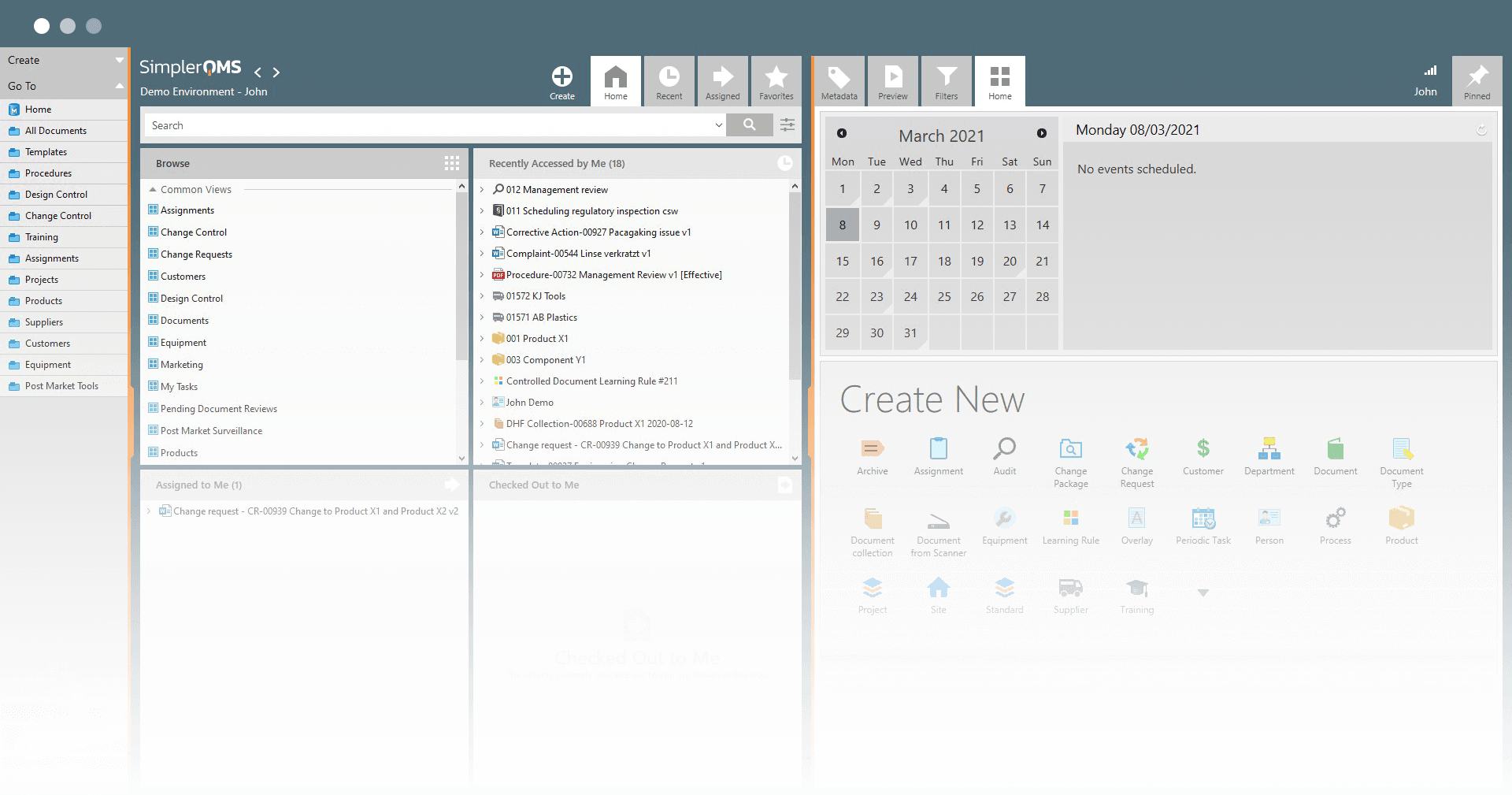
Lead Digital Transformation in Your Organization
Empower your organization with the tools they need to get more work done, faster, in compliance with the regulatory requirements.
TRUSTED BY
























challenge
Complex Implementation and Integration
A good QMS software solution should have the flexibility to integrate with your current systems and applications.
Ideally, you want to choose a vendor that fits your current infrastructure, this way you minimize the risk, uncertainty, and amount of work when implementing the system.
solution
Microsoft Based Infrastructure
SimplerQMS can easily be connected to Microsoft Entra ID (previously known as Microsoft Azure Active Directory) for single sign-on and user data management.
The system allows you to work with all the familiar Office applications like Word, Excel, and PowerPoint, thanks to its integration with Microsoft Office 365.
Documents are simply edited in the Microsoft Office interface and saved with one click in SimplerQMS. This means you don’t have to download, edit, and upload the document every time you make a change.
solution
Seamless Integration With Other Applications
SimplerQMS can easily be integrated with other systems like Enterprise Resource Planning (ERP), Customer Relationship Management (CRM), Product Lifecycle Management (PLM), Warehouse Management System (WMS).
We also provide an application programming interface (API). The API makes it possible to access SimplerQMS from within scripting environments. This means you can integrate SimplerQMS with many other applications.
challenge
Lack of Security and Regular Backups of Data
Undoubtedly, one of the biggest concerns in any IT department is the security of the data.
Data must be encrypted, and data backups need to be regular.
Servers should also be regularly updated and maintained by qualified professionals to assure steady performance.
solution
Trusted Technology Platform With M-Files
SimplerQMS is built on robust and disaster-proof technology by leveraging the M-Files Document Management platform as the core of its system.
M-Files technology platform serves over 5,000 customers across 100 countries. It is well developed and tested to reach the highest levels of reliability and efficiency.
solution
Secure and Scalable Hosting With Microsoft Azure Cloud
SimplerQMS uses the highly secure Microsoft Azure Cloud Storage solution. Servers are regularly updated and maintained by qualified professionals to assure steady performance.
This guarantees an up-to-date, reliable service with no additional workload on your side.
challenge
Issues With Regulatory Compliance
You do not want to put the responsibility of meeting the specific compliance requirements of the QMS software and its processes on yourself, such as Computer Software Validation and requirements for Electronic Signatures.
solution
Full Regulatory Compliance
SimplerQMS offers a software solution that complies with FDA 21 CFR Part 11 and EU GMP Annex 11 regarding the requirements for electronic signatures, electronic records, and computerized systems.
SimplerQMS supports compliance with Life Science requirements, such as ISO 9001, GxP, ISO 13485, FDA 21 CFR Part 210, 211, and 820, ICH Q10, EU MDR and IVDR, and others.
Our cloud storage and technology platform providers, Microsoft Azure Cloud and M-Files, also adhere to the necessary requirements for the life science industry, such as ISO 9001, ISO/IEC 27001, and ISO/IEC 27018.
solution
Continuous System Validation
SimplerQMS fulfills the Computer System Validation requirement for Life Science companies that digitally store and manage quality records.
The system is fully validated according to ISPE GAMP5 and undergoes revalidation for each new version or standard update. SimplerQMS complies with requirements regarding the validation of systems used for QMS such as 21 CFR Part 11, and 820, EU GMP Annex 11, and ISO 13485:2016.
This means that your IT department does not have to spend any time or resources on software validation. We do all the software validation for you, eliminating any additional expenses, resources, or time commitments on your part.
modules
The SimplerQMS Modules Cover All Your Processes
No matter if your life science organization is a start-up or a later-stage company, our integrated modules cover your needs. All of the modules are included in the SimplerQMS subscription.
Training Management
Save time with automated training activities, learning overview, reminders, and generation of training certificates.
CAPA Management
Identify, uncover, resolve, and report all the preventative actions and corrective actions (CAPAs) seamlessly.
Complaint Management
Reduce the associated risks and resolve issues quickly by optimizing complaint management processes.
Change Management
Recognize and manage all changes accordingly to ensure compliance and structure within your organization’s QMS.
Design Control
Manage all the necessary processes related to product design and meet design control requirements with ease.
Document Control
Automate and standardize your document control activities with ease.
Risk Management
Consolidate risk and handle your risk management file in a well-organized and structured manner.
Supplier Management
Simplify supplier-related activities and handle your supplier documentation following the standards.
See SimplerQMS in Action
To see SimplerQMS in action and learn how you can make the most of it, request a personalized demo presentation.