Supplier Quality Management Software for Life Sciences
Qualify and monitor suppliers to maintain oversight, traceability, and regulatory compliance.
Book a DemoJoin Over 5,000+ Quality Professionals Who Trust SimplerQMS
Comprehensive Supplier Quality Management Within a Complete eQMS
SimplerQMS Supplier Quality Management Software solution helps you manage all supplier-related quality processes in a single system.
SimplerQMS provides a supplier management module that is part of a complete eQMS solution, which includes all core QMS modules such as document control, training management, change management, nonconformance management, CAPA management, and more.
The following are some of the core capabilities of our supplier management software solution.
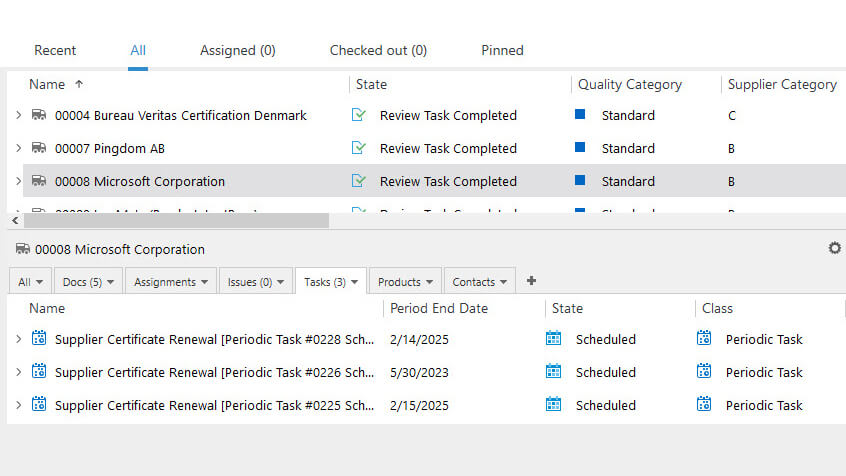
Evaluate, Qualify, and Manage Your Suppliers
Streamline supplier qualification, performance evaluations, and subsequent supplier performance monitoring using pre-configured workflows based on Life Science requirements.
Manage supplier qualification process from start to finish – select the responsible persons in the workflow, define the supplier category, perform qualification tasks, and manage reviews.
Maintain your approved supplier lists (ASL), and use document templates to create records for contracts, surveys, evaluations, certificates, incoming inspections, and more. Manage all your supplier records in an integrated cloud-based system. Easily define access levels to prevent unauthorized user access.
Streamline Supplier Audit Processes
Supplier audits are an important part of supplier quality management but can be time-consuming and resource intensive.
SimplerQMS simplifies planning, managing, and tracking of supplier quality audits. Create supplier audit plans and have relevant people automatically notified by email about their assignments and due dates.
Utilize customizable views and have all supplier-related data readily available. Also, during the audit situation when you need to show the relevant evidence to inspectors and auditors quickly.


Overview Status of Your Suppliers
Schedule supplier requalifications, reviews, assignments, and periodic tasks and have an overview of all processes.
Relate supplier documents to products, departments, customers, non-conformances/deviations, CAPAs, supplier quality issues, audit plans, and other relevant files while drafting your document with just a few clicks.
Drag and drop files and emails to suppliers to store records in one location, which is accessible for relevant employees and during audits.
Export data from your views to further analyze trends, such as the number of qualified suppliers, the number of supplier-related issues, suppliers with periodic tasks pending, and more.
Integrate With Other Quality Processes
Different tools for different quality processes can make it difficult to get a holistic view of supplier quality performance.
SimplerQMS supplier quality management software integrates all core quality processes, such as document control, training, change management, CAPAs, audit management, and more.
For example, you can easily create Supplier Corrective Action Reports (SCAR) directly from a non-conformance/deviation and have all documents interlinked.


Connect to Other Software Applications
Reduce data silos by connecting all software in your organization.
Integrate SimplerQMS with your existing applications, such as ERP or SCM solution, using an open API.
Ensure Compliance With Regulatory Standards
SimplerQMS provides features that support supplier quality management processes and help you achieve compliance with regulatory requirements, such as FDA cGMP, ISO 13485:2016, FDA 21 CFR Part 820, ISO 9001:2015, and others.
Pre-configured workflows based on life science requirements allow you to define the supplier category, create supplier audit plans, and more.
FDA 21 CFR Part 11 and EU Annex 11 compliant electronic signatures time-stamped audit trails are included to ensure complete traceability of your QMS.

What Customers Achieve With SimplerQMS
“It’s very flexible, smooth, and easy to use. Documents no longer get lost and the whole history of all products is accessible for anyone at any time.”
Discover How SimplerQMS Can Help You
Complete eQMS Software for Life Sciences
Frequently Asked Questions
Supplier Quality Management (SQM) is a process that enables organizations to ensure that their suppliers meet all the requirements and provide products of the required quality.
It involves the creation of approved supplier lists (ASLs), setting internal criteria for suppliers, qualification of suppliers, assessment of supplier performance, auditing suppliers, and so on.
Supplier Quality Management software is a tool that helps organizations to streamline the management of their suppliers and ensure that they meet all the requirements.
It enables organizations to qualify and categorize suppliers, assess their performance, and plan supplier audits to check for conformity.
Features like automated document routing, notifications, reminders, customizable supplier status views, monitoring reports, and others, make it easier for companies to manage supplier-related processes.
Some of the most popular SimplerQMS Supplier Quality Management software features include the following:
Integrated and cloud-based system. Easily store, share, and manage all your supplier-quality documents in one system.
Electronic signatures. FDA 21 CFR Part 11 and EU Annex 11 compliant electronic signatures make it easy to get sign-offs on documents and track who has signed them.
Time-stamped audit trails. All actions taken in the software are time-stamped and auditable to ensure complete traceability of your supplier quality management system.
Microsoft Office integration. Work with your documents in the familiar Microsoft Office interface using applications like Word, Excel, and PowerPoint.
Integration with other quality processes. Interconnect your supplier quality management system with other quality processes, such as document control, training, change management, non-conformances, audits, CAPA, risk management, and others. Plus, you can integrate SimplerQMS with your ERP solution through an open API for a complete solution.
Pre-configured workflows. Use integrated workflows based on Life Science requirements to save time and ensure compliance.
The total cost of SimplerQMS depends on the type and the number of licenses you purchase.
SimplerQMS supplier quality management software is part of a complete QMS solution, which includes all QMS modules, implementation, training, ongoing support, validation, hosting, and more, for one subscription price.
This means that everything is included in the price, and there are no other costs associated with subscribing to SimplerQMS.
Please visit our pricing page if you are interested in learning more about the license type and the many features and services included.
SimplerQMS Software typically takes five to six weeks to be implemented.
Supplier management is integral to our eQMS solution and can be implemented after document control, change management, and training management modules.
Implementation times may vary depending on the number of documents that need to be created and/or migrate and the company’s resources.
