Document Control for Life Science Companies
Document Control in SimplerQMS helps Life Science companies manage controlled documents with structured workflows that support regulatory requirements.
Book a DemoTrusted by 5,000+ life science professionals worldwide – and growing.
15,000+ life science professional community
4.6 rating on Capterra
4.9 rating on G2
Case study
Case study
Case study
Case study
Case study
Explore Document Control in SimplerQMS
Disconnected systems, manual processes, and outdated documents put compliance at risk.
See how SimplerQMS centralizes document control, streamlines workflows, and ensures only current, approved versions are in use in this guided product tour.
What You Can Achieve with SimplerQMS
Built by life science experts to simplify document control while maintaining compliance and traceability.
“One of the positive outcomes for our organization and operational efficiency is that the document control team works much more effectively since we have implemented SimplerQMS.”
Improved Traceability and Automation
Centralize document control and replace manual routing with automated workflows. Improve traceability and reduce manual work.
Confident Compliance
Designed to support alignment with key regulations and standards such as FDA 21 CFR Part 11, ISO 13485, and EU GMP Annex 11. Helps maintain audit-ready documentation.
Data-Driven Decisions with Built-In Analytics
Monitor document activity, compliance metrics, and operational KPIs in one place. Make more informed decisions with clearer visibility.
Faster Implementation with Included Support
Go live in 6–8 weeks with structured implementation, training, and 24/7 support. Reduce disruption and support faster adoption.
How Different Roles Benefit from Document Control in SimplerQMS
Life science teams use SimplerQMS to support quality, regulatory, and operational responsibilities.
Quality Assurance

Regulatory Affairs

Research and Development (R&D)

Manufacturing and Operations

Management and Executives

How Life Science Teams Use Document Control in Practice
Life Science organizations rely on SimplerQMS to manage document control processes efficiently while ensuring compliance, traceability, and audit readiness.
Maintain Controlled SOP Across Quality Processes
SimplerQMS helps teams maintain consistent SOP control, support audit readiness, and reduce manual coordination across teams.
Control Documents Across Quality and Manufacturing
SimplerQMS helps teams maintain controlled documentation across the product lifecycle while supporting key Life Science requirements.
Prepare for Audits with Confidence
SimplerQMS helps teams stay audit-ready with complete, traceable documentation and easier access to records during inspections.
Maintain Controlled Documentation Across Projects
SimplerQMS helps organizations maintain consistent, controlled documentation across multiple projects while supporting compliance.
How SimplerQMS Supports These Use Cases
Explore the key capabilities in SimplerQMS that support document control and help Life Science teams manage this process in a structured and controlled way.
Automated Workflows
Guide documents through draft, review, approval, and effective stages with less manual follow-up.

Electronic Signatures
Capture traceable approvals with electronic signatures designed to align with FDA 21 CFR Part 11 requirements.
Learn about electronic signatures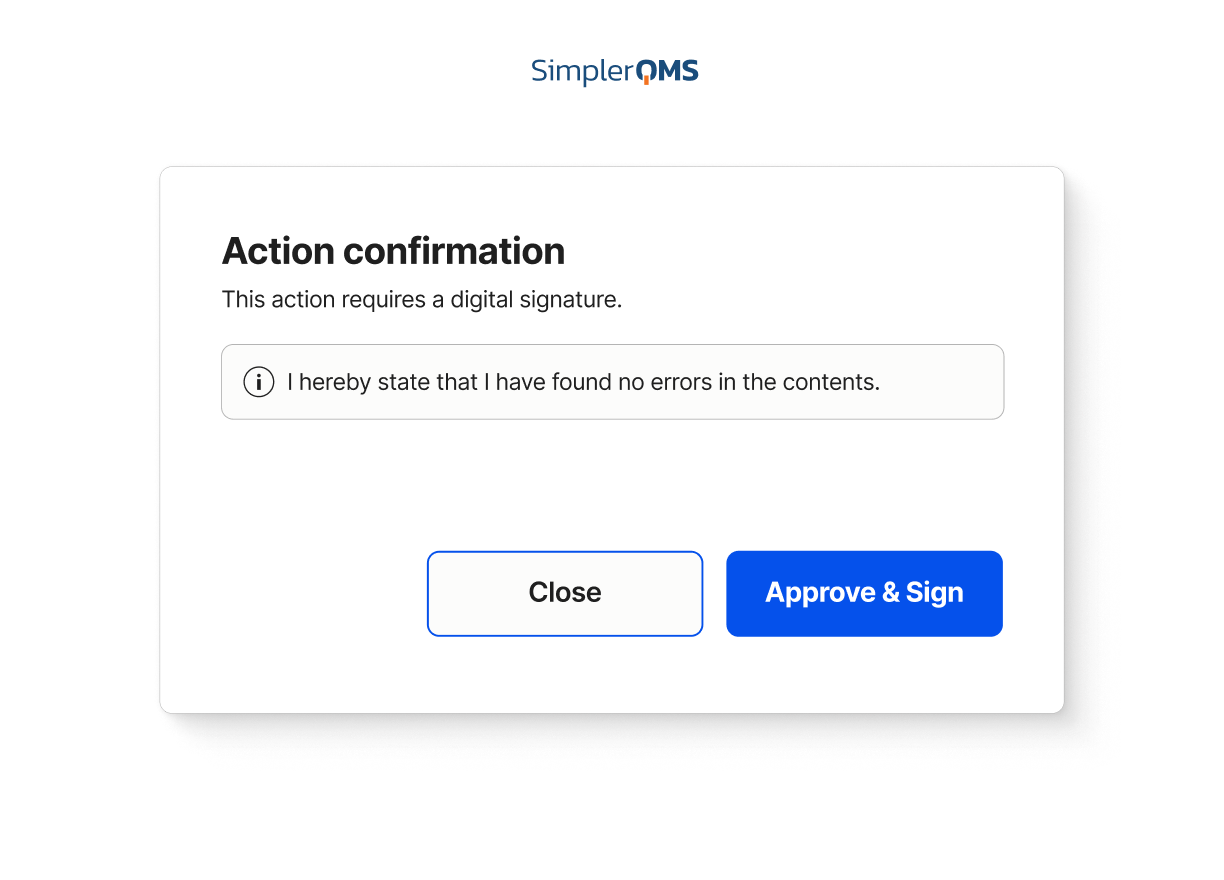
Generate Summary (AI-Powered)
Generate change summaries automatically to support faster review and more consistent documentation.
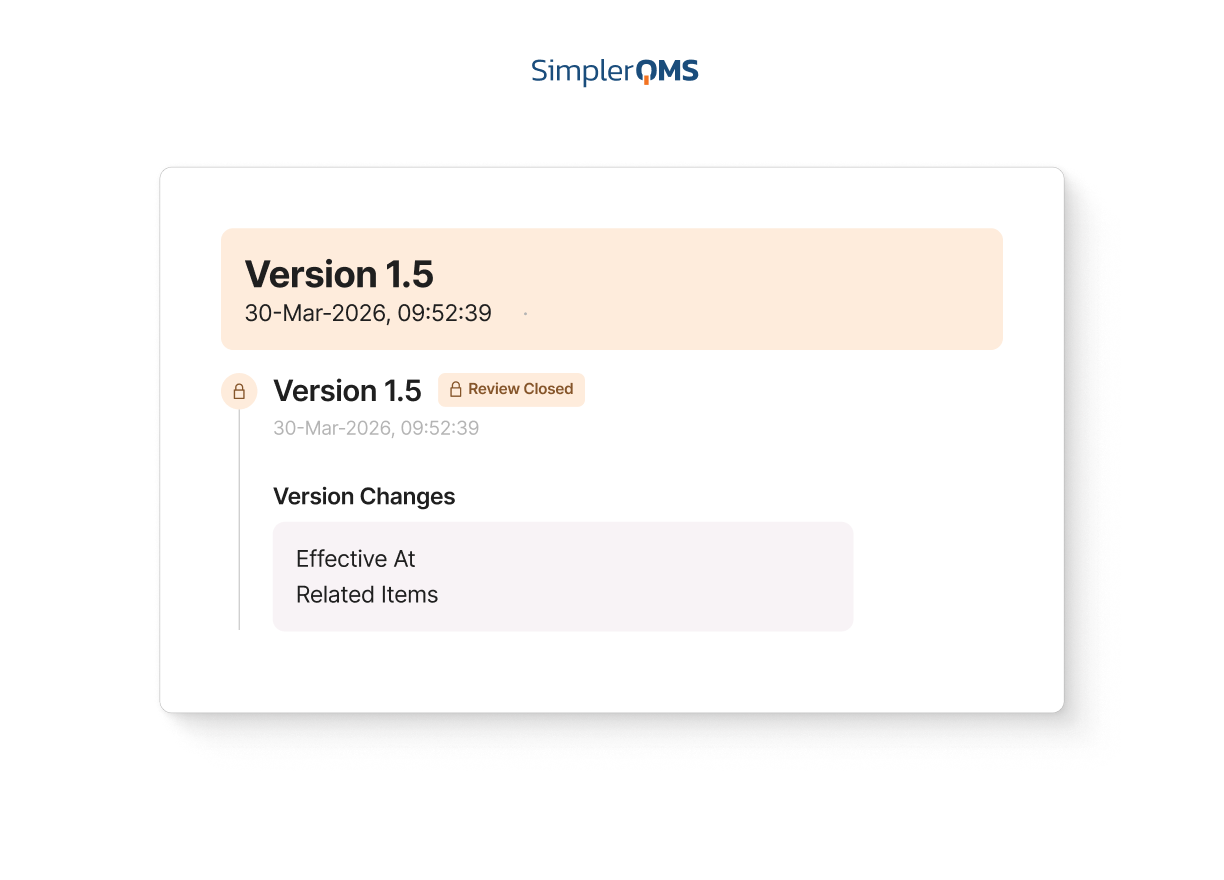
Version Control and Document History
Help teams work from the latest approved document with clear version history and traceability.
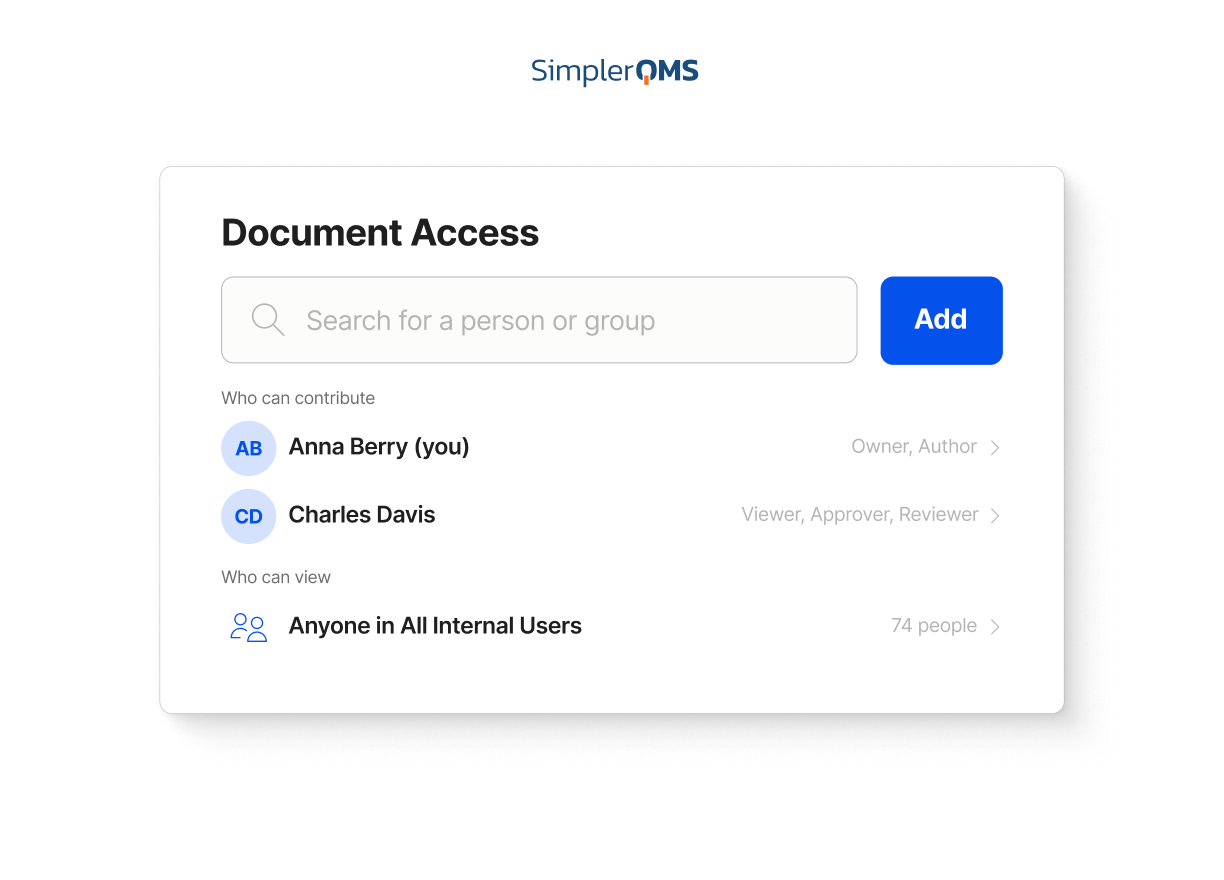
Role-Based Access Control
Control who can view, edit, review, or approve documents based on role.
Document Properties and Classification
Classify documents by type, process, owner, and effective date for better control and searchability.
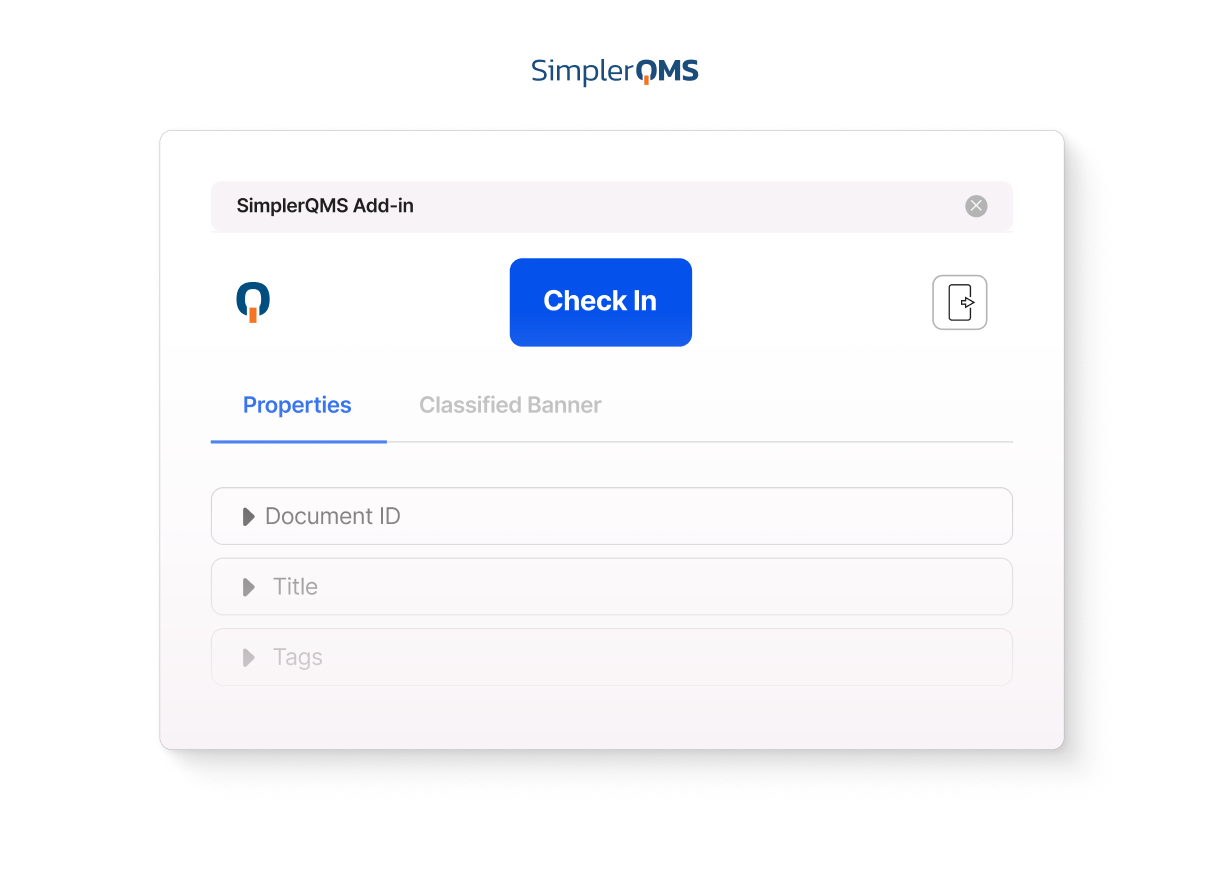
Microsoft Office Integration
Draft and edit documents in Microsoft Word while keeping document control in SimplerQMS.

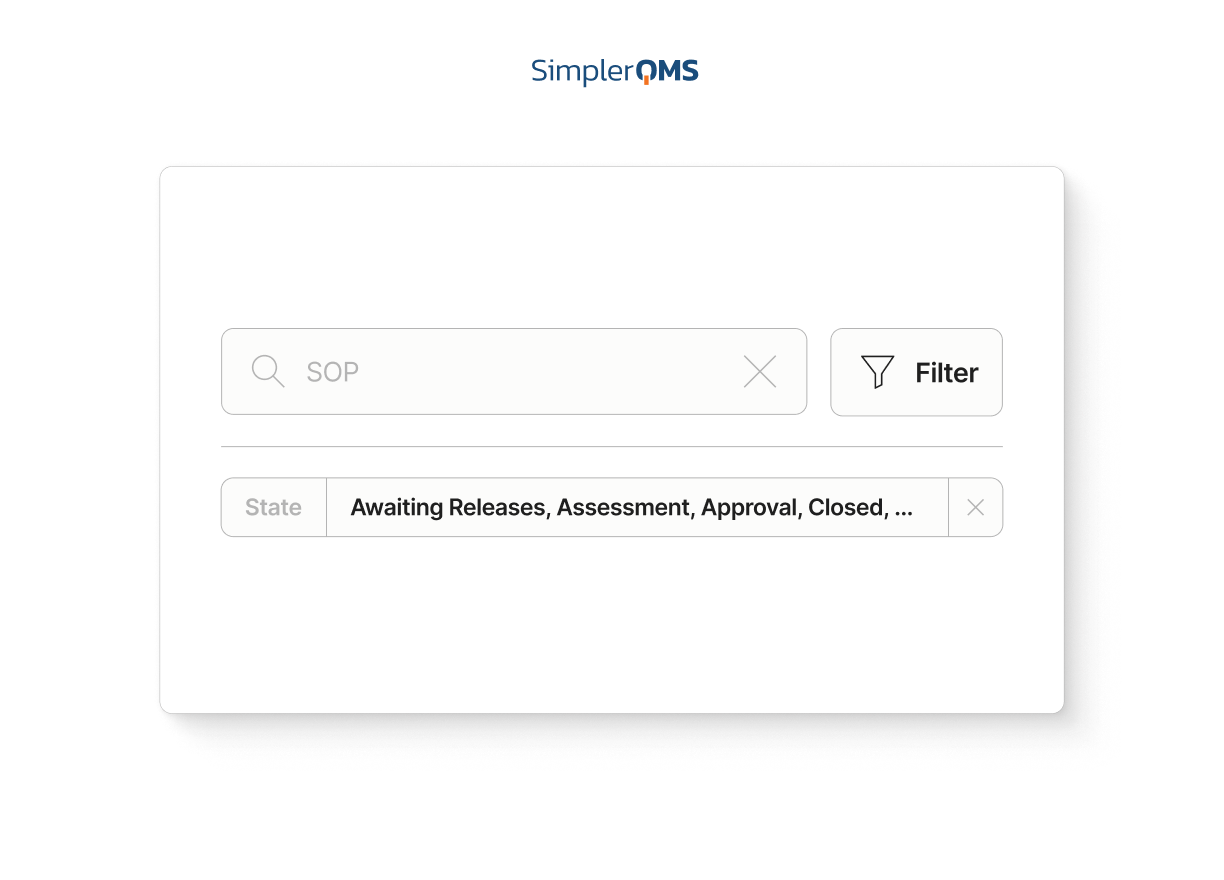
Search, Filtering, and Easy Navigation
Find controlled documents faster using search, filters, and document properties.
Document Control is Built into an Integrated eQMS
Document Control in SimplerQMS is part of an integrated eQMS for Life Science companies. It connects documents with related quality processes to support consistency, visibility, and compliance.
Explore our QMS Software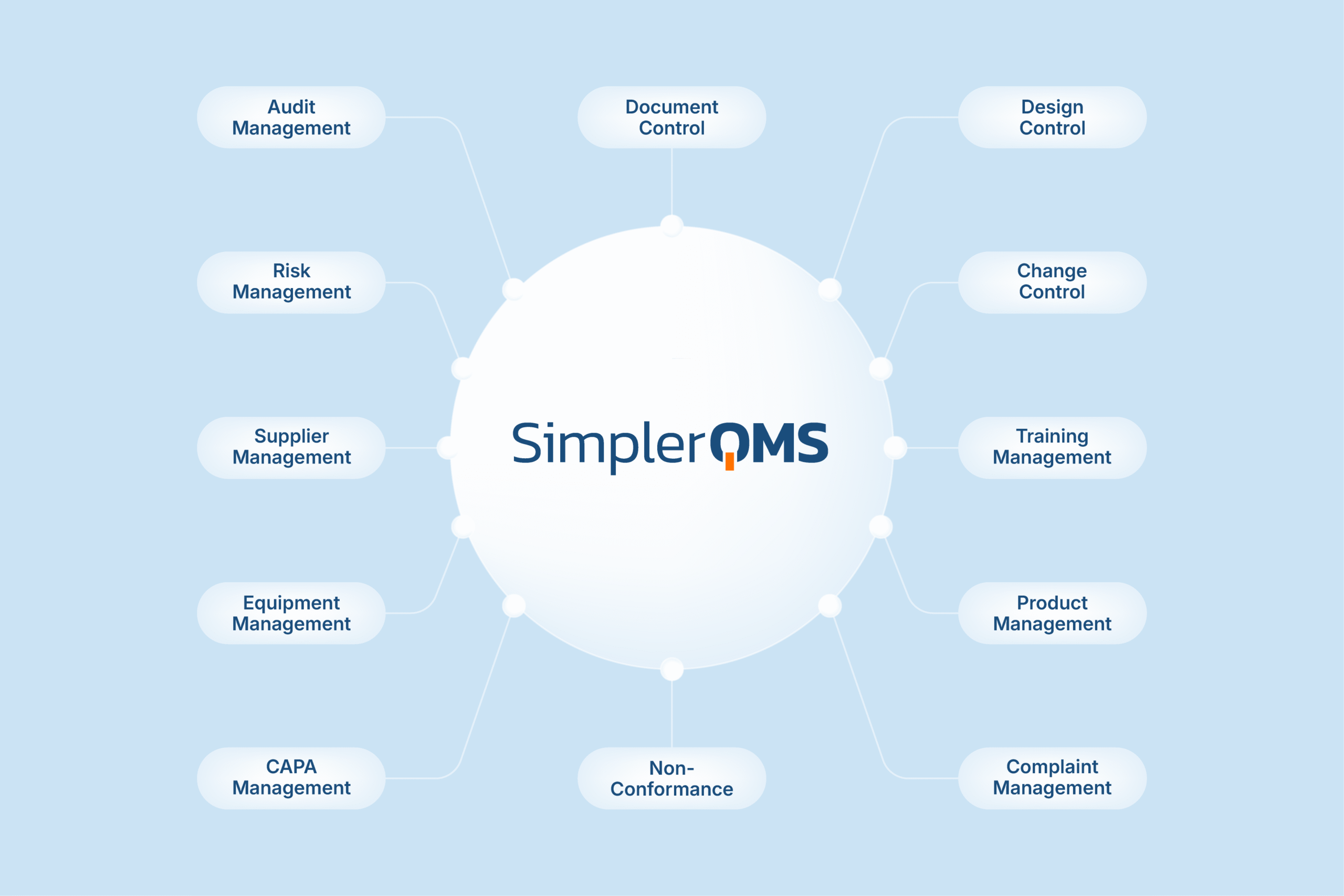
More Than Just Document Control
Beyond document control, SimplerQMS supports a wide range of interconnected quality processes within a fully integrated eQMS.
Supporting Compliance with Life Science Requirements
SimplerQMS enables organizations to implement document control processes aligned with key Life Science requirements by helping organizations maintain controlled processes, traceability, and documented oversight across the document lifecycle.
Hear From Our Happy Customers
Explore real-world case studies and customer reviews that show how Life Science teams use SimplerQMS.
‘’SimplerQMS has been an excellent solution for our team. The platform is intuitive and user-friendly, and the implementation process was incredibly smooth. We were able to go live in just 6 weeks…’’
“The ease of use, customizability, and the support given by the technical support team…Very fast in their response.”
‘’SimplerQMS is much better than our previous paper-based QMS system…SimplerQMS gave us excellent pricing, customer support for understanding how to use their system and set up our QMS and is easy to use.’’
“…I have consistently been impressed with its flexibility and range of features. These features have met all my needs in every aspect. The excellent support team further enhances the overall experience.”
“Amazing customer support and help. The system is clearly structured. After the introduction, it is easy to use.”
‘’User-friendly and simple to use. Easy to customize to fit your individual needs.’’
‘’Exceptional customer support! Compliant to 21 CFR Part 11 and ISO 13485:2015.’’
‘’…SimplerQMS offers the bare necessities and is perfect for our needs.’’
Explore Document Control Resources
Explore practical insights and guidance on document control in regulated life science environments.

SOPs in the Pharmaceutical Industry: Definition, Types, Format, Guidelines, and Management
Document ControlPharmaceutical
Medical Device Document Control: Definition, Process, and Requirements
Document ControlMedical Devices
Pharmaceutical Document Management: Definition, Requirements, and Software
Document ControlPharmaceutical
Frequently Asked Questions (FAQ) about Document Control
Find answers to common questions about document control and its role in life science quality management.
What Is Document Control?
Document control is the process of creating, reviewing, approving, distributing, updating, and retiring controlled documents such as SOPs, policies, and work instructions. In Life Science environments, document control helps ensure that only approved and current documents are used and that changes are traceable.
Why Is Document Control Important for Life Science Companies?
Document control supports consistency, traceability, and oversight across quality processes. It plays a key role in audit readiness and helps reduce risks related to outdated, incomplete, or uncontrolled documentation.
How Is Document Control Supported in SimplerQMS?
SimplerQMS supports document control through structured workflows, defined document properties, version control, and controlled lifecycle states. Documents move through draft, review, approval, effective, update, and retirement in a system-supported way.
Can SimplerQMS Support Electronic Signatures for Document Approval?
Yes, SimplerQMS supports electronic signatures for document approvals as part of the document control process. Approval actions are recorded with user credentials and timestamps to support traceability.
How Does SimplerQMS Help With Version Control and Traceability?
SimplerQMS maintains document version history by separating working copies from effective versions. Each update follows a controlled process, helping teams track what changed, when it changed, and who approved it.
How Does Document Control Connect to Other Quality Processes in SimplerQMS?
Document control in SimplerQMS is supported within an integrated eQMS. Controlled documents can be linked to related processes such as training, change activities, or audits, helping maintain continuity and visibility across the quality system.
See SimplerQMS in Action
See how SimplerQMS supports document control and other interconnected quality processes in a single, integrated eQMS.
Book a Demo