Regulatory Document Management Software (rDMS) for Life Sciences
Manage regulatory documents in a controlled environment to support traceability and submission readiness.
Book a DemoJoin Over 5,000+ Quality Professionals Who Trust SimplerQMS
Streamline Regulatory Document Management in Your Organization.

Ensure a Unified Access to All Regulatory Documents
Effective document control is essential to any regulatory environment, as it helps organizations achieve efficiency in their operations while maintaining compliance.
With SimplerQMS regulatory document management software (rDMS) you can store all your regulatory documents in one centralized place for easy and quick access by authorized personnel with the appropriate permissions.
Search for documents using an advanced “Google-like” search feature and find the right document easily, even if you don’t know its exact location. Plus, cloud-based deployment provides users with access to the system from anywhere, at any time.
Facilitate an Entire Regulatory Documentation Lifecycle
Take advantage of automated workflows for the creation, review, approval, distribution, escalation, and archiving of regulatory documents in a complaince and efficient manner.
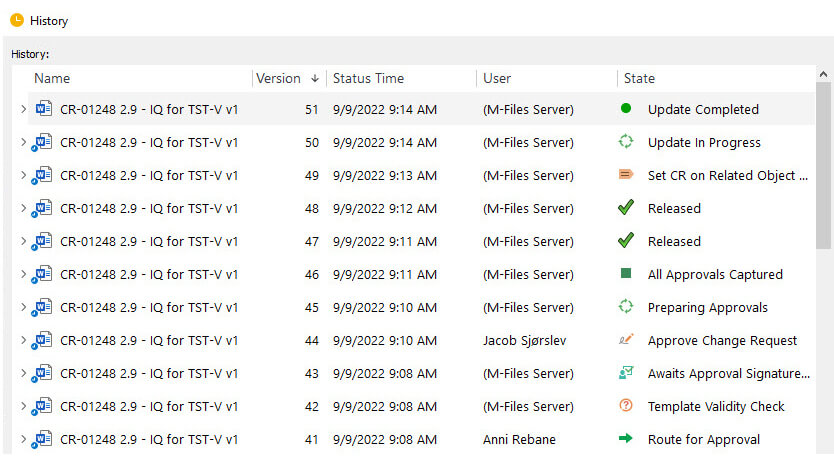
Assign documents to specific users or groups for review and approval, track the status of each document, and receive notifications and reminders when a task is due. Track document versions, revision histories, and document status throughout the entire process to ensure that everyone is always working on the most up-to-date version of a document.
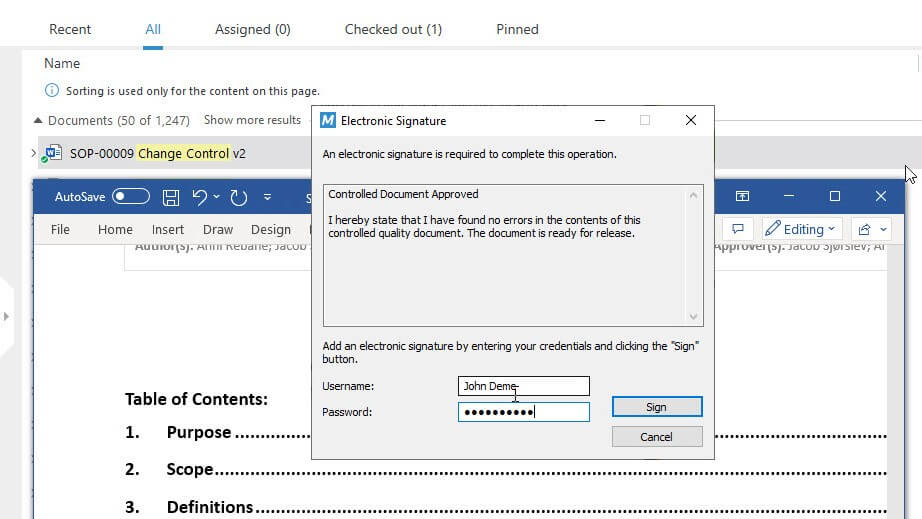
Furthermore, SimplerQMS allows you to access full, time-stamped audit trails, create documents based on predefined templates, edit documents in familiar Microsoft Office applications, and approve documents using eSignatures.
Meet Complex Regulatory Requirements With Ease
SimplerQMS is purposefully built to meet the specific needs of highly regulated life science industries such as medical device, pharmaceutical, biotechnology, and others.
The software helps manage documents related to quality management, clinical operations, regulatory affairs, and more, in a secured and controlled manner to ensure compliance with FDA regulations, ISO standards, and other requirements.
Moreover, SimplerQMS provides a fully 21 CFR Part 11 compliant document management solution, which means that it meets the stringent requirements for electronic records and signatures set forth by the U.S. Food and Drug Administration (FDA).
Combine Regulatory and Quality Data in a Single Platform for Greater Efficiency
In today’s fast-paced and ever-changing regulatory environment, life science organizations need to be able to move quickly and efficiently.
With SimplerQMS you can manage both RA and QA documents in one system and get a complete picture of your organization’s compliance posture. We provide a broad QMS process support, so you can not only manage regulatory documents, but also quality control plans, NCs and deviations, CAPAs, suppliers, employee training, audits, and more.
Plus, you can integrate SimplerQMS with third-party software applications such as your ERP, CRM, or LIMS system through our API to further streamline your business processes.

eQMS That Goes Beyond Document Management
Frequently Asked Questions
Regulatory document management software (rDMS) is a software application designed to help life science organizations to manage regulatory documents.
The software enables users to author, review, approve, distribute, and archive documents in a controlled, efficient, and secure manner. Regulatory DMS software allows organizations to streamline their regulatory document lifecycle from creation to submission and obsolescence while ensuring compliance with regulatory authorities.
There are many benefits of using Regulatory DMS software including improved compliance with regulatory authorities, faster document lifecycle times, greater efficiency and productivity, reduced risks associated with manual processes, and more enhanced security and control over documents.
Popular features of SimplerQMS Regulatory Document Management Tools include:
- Controlled document workflows
- Version controls and document history
- User access controls and permissions
- Full audit trail of all document changes
- Centralized, cloud-based, repository with easy search and retrieval
- FDA 21 CFR Part 11 compliant electronic signatures
- Broad QMS process support
- Integration with third-party software applications
- Access to documents from any device
All of SimplerQMS’s software tools are pre-validated and compliant with the requirements of GxP Guidelines, FDA 21 CFR Part 11, and ISO 13485:2016.
We also provide extensive validation evidence for use during audits and inspections. This includes our validation plan, procedure, report, as well as, IQ, OQ, and PQ documentation.
Plus, we ensure continuous re-validation of our software tools to maintain compliance which is something many other software providers do not offer.
SimplerQMS’s regulatory document management is part of an all-in-one QMS software solution. This includes all QMS modules (document control, change control, audits, NCs, CAPAs, suppliers, training, etc.), system implementation, training, ongoing support, continuous re-validation, hosting, and more.
This means that everything is included in the price you pay and there are no other costs associated with subscribing to SimplerQMS.
To learn more about our pricing, please contact us for a demo and price quote.
What Customers Achieve With SimplerQMS
“It’s very flexible, smooth, and easy to use. Documents no longer get lost and the whole history of all products is accessible for anyone at any time.”
